Thermodynamics
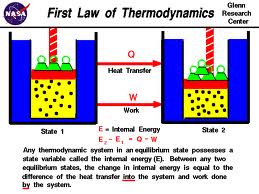
Thermodynamics is the study of conversions between thermal energy and other forms of energy. Energy cannot be created or destroyed,but it can be converted into different forms. Thermal energy flows spontaneously hotter objects to colder ones. The first law of Thermodynamics states What must happen for thermal energy to flow from a colder object to a hotter object? Only if work is done on a system according to the second law of thermodynamics. Waste Heat is thermal energy that is not converted into work.The third law of Thermodynamics states that absolute zero cannot be reached. The lowest temperature recorded is three billionths of a kelvin above absolute zero.